|
This portion of the trial is now enrolling patients. On January 9, 2023, 4DMT disclosed that the Company had initiated the randomized Phase 2 portion of the Phase 1/2 PRISM clinical trial for 4D-150 in patients with wet AMD. Initial Cohort 1 data (n=5) from the Phase 1 portion of the Phase 1/2 PRISM clinical trial with 4D-150 for wet age-related macular degeneration (wet AMD) demonstrated a reduction in annualized anti-VEGF injection rate by over 95%, further validating the potential of our intravitreal R100 vector for other large market eye diseases such as geographic atrophy. The IND clearance enables the initiation of SPECTRA clinical study sites, and 4DMT expects to begin enrollment in the third quarter of 2023. The doses to be evaluated in DME are anticipated to be between 6E9 to 3E10 vg/eye. 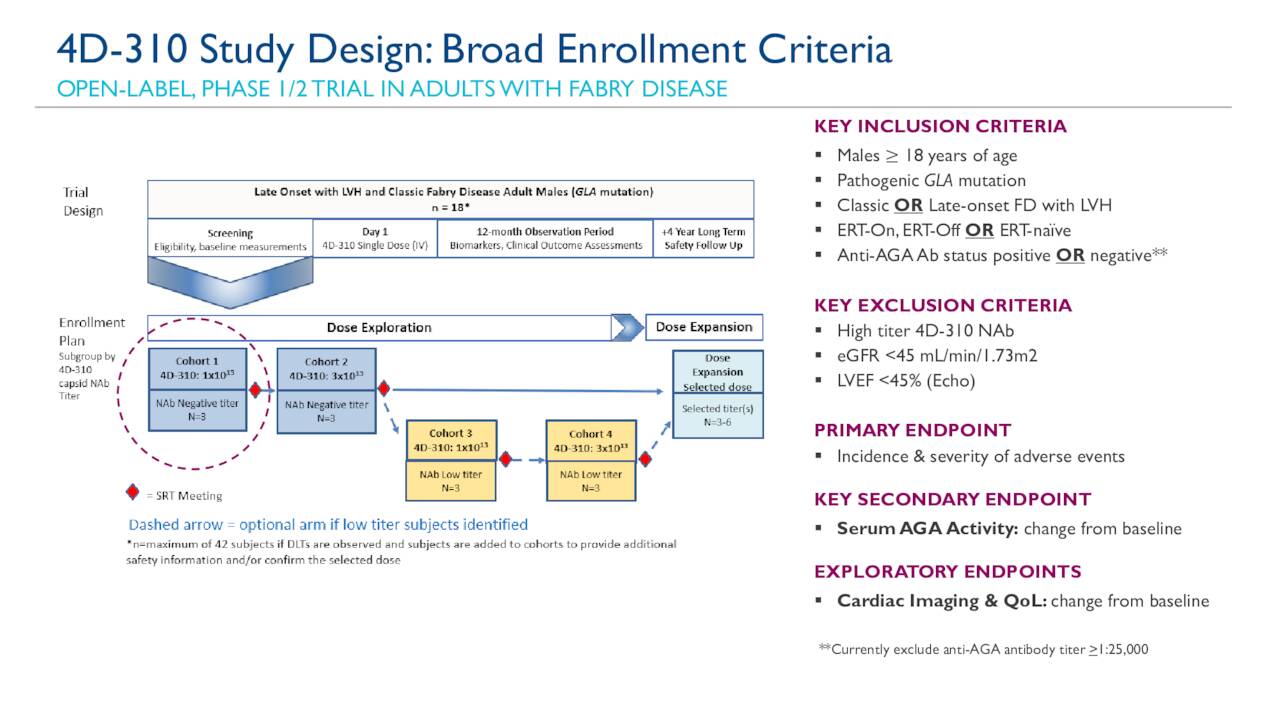
The study design consists of a Dose Confirmation stage followed by a masked Dose Expansion stage, in which patients will be randomized to receive a single intravitreal injection at one of two dose levels of 4D-150 or aflibercept in a 1:1:1 ratio (n=54 patients). The Phase 2 SPECTRA clinical trial will assess 4D-150 in patients with DME. 02, 2023 (GLOBE NEWSWIRE) - 4D Molecular Therapeutics (Nasdaq: FDMT, “4DMT”), a clinical-stage biotherapeutics company harnessing the power of directed evolution for targeted genetic medicines, announced FDA clearance of the Investigational New Drug Application (IND) for 4D-150, an R100 vector-based intravitreal genetic medicine, for the treatment of patients with Diabetic Macular Edema (DME). Randomized Phase 2 portion of the Phase 1/2 PRISM clinical trial with 4D-150 for wet AMD is currently enrollingĮMERYVILLE, Calif., Feb. Interim PRISM data for dose Cohorts 1, 2, & 3 (n=15) to be presented at the 2023 Association for Research in Vision and Ophthalmology (ARVO) Annual Meeting Initial Phase 1 PRISM clinical trial with 4D-150 for wet age-related macular degeneration further validates R100 intravitreal vector potential for other large market eye diseases including geographic atrophy MedImmune has been signing a number of research agreements of late, as it looks to build its pipeline of products, and entered a five-year collaboration with Ethris looking into respiratory diseases in August 2017.Įarlier this year it entered a partnership with Compugen for multiple bi- and multi-specific antibodies, though the particular therapeutic area was not revealed.4D-150 Phase 2 SPECTRA clinical trial for diabetic macular edema enrollment is expected to initiate in Q3 2023 MedImmune had not responded to a request for further information on the exact condition that will be targeted or on the financial details of the deal at the time of publishing. Our progress in customised AAV vectors enables us to unlock the potential of gene therapy and, with MedImmune’s expertise in protein engineering, we will continue to push boundaries in proprietary gene delivery to tissues and cells.” In a joint statement, David Kirn, CEO and co-founder of 4DMT, said: “This exciting collaboration may open the door to significant advancements in treatment for respiratory patients. This would allow any potential therapy to bring a ‘blueprint’ for the cell to produce a protein, which could potentially interrupt or alleviate symptoms of chronic lung disease.Īs part of the partnership, 4DMT will work on discovery, engineering and optimising the development process, while MedImmune will work on the clinic development of any therapeutic candidate discovered.  
AAV vectors are designed to transport genes into both dividing and nondividing cells without introducing genetic material into the host genome. The target for the gene therapy will be chronic lung disease. MedImmune, AstraZeneca’s research and development arm, has opted to work with California-based 4D Molecular therapeutics (4DMT) on its adeno-associated virus (AAV) gene therapy vector discovery and engineering program.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
